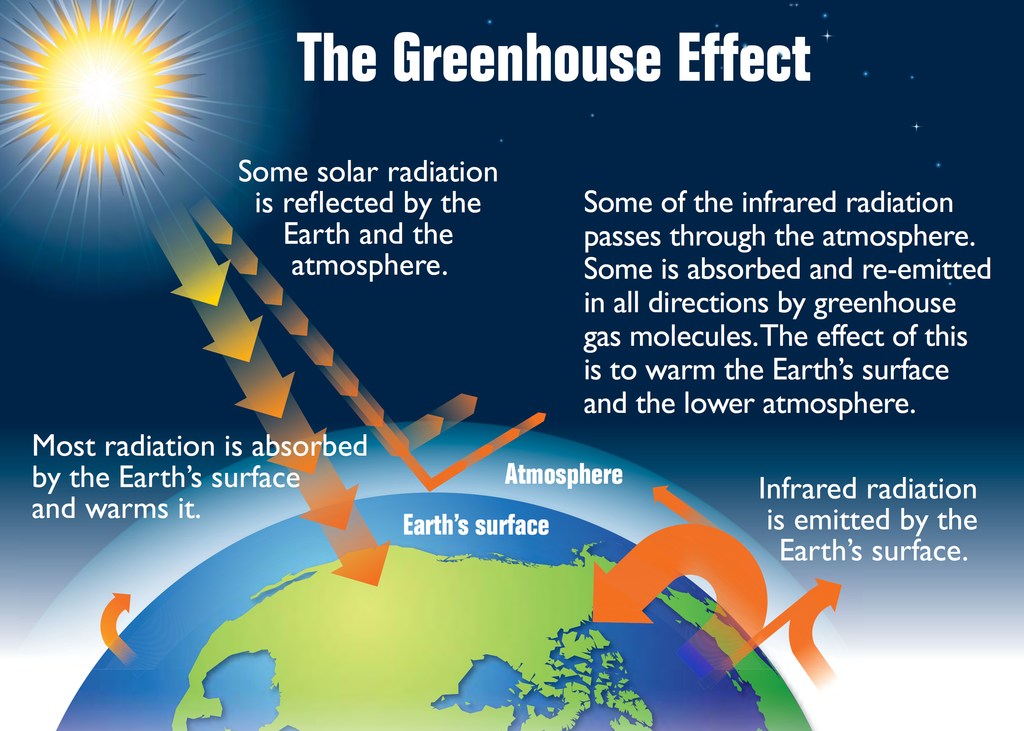
What is the Greenhouse Effect?
Definition of the Greenhouse Effect
“When we burn organic (i.e. carbon-containing) fuels, or organic matter decomposes, carbon dioxide is released into the air. It is transparent to incoming solar radiation, but opaque to some wavelengths of heat radiated from the warmed surface of the Earth, and so traps heat, leading eventually to a warming of the lower atmosphere” ((Allaby, M. Green Facts. London: Reed, 1989. Print)).
This is known as the greenhouse effect, as in principle, the atmosphere behaves in a similar manner to a garden greenhouse – it allows sunlight to penetrate, but heat is trapped within the atmosphere in the same way that it is trapped within the glass walls of a greenhouse. This trapped hot air is causing the earth to heat up, resulting in global warming, and ultimately climate change.
Contents
Is it Natural, or Caused by Humans?
Greenhouse gases include naturally occurring gases, such as carbon dioxide, methane, and even water vapor. In fact water vapor is the most abundant greenhouse gas. However human activities, such as burning of coal, exhaust fumes from vehicles, and burning of trees during deforestation activities, are contributing huge amounts of additional greenhouse gases into the atmosphere, where they enhance the greenhouse effect further and contribute to global warming.
Greenhouse Feedback Mechanisms
As the Earth warms up it triggers other impacts, such as ice sheets to melt, which amplify the greenhouse effect and feedback into the cycle of warming, causing the Earth to get even hotter still.
The Albedo Effect
Snow capped mountains and ice sheets reflect radiation away from the Earth – a phenomenon that is known as the albedo effect. This helps reduce the amount of heat absorbed by the Earth, and therefore plays a vital role in keeping the Earth cool.
When ice sheets melt, it exposes dark rock or vegetation that doesn’t have the same reflective properties as ice, and thus tends to absorb rather than reflect heat.
Greenhouse Gases Released
As sea ice and ice covering the Tundra melts, it exposes methane hydrates – ice crystals that contain pockets of methane locked within. When methane hydrates are exposed to warm air or warm water they begin to melt, releasing the methane – a greenhouse gas that is more than 20 times as potent as carbon dioxide in terms of its warming effect.
Furthermore, as the atmosphere heats up, so do the oceans, which leads to more water being evaporated into the atmosphere. Consequently there is more water vapor present in the atmosphere. This not only contributes to severe weather, but because water vapor is a greenhouse gas, it also contributes to further warming. Are you beginning to understand the knock-on effects?
Other factors of influence
How latitude impacts magnitude of the greenhouse effect
According to the IPCC ((https://www.ipcc.ch/publications_and_data/ar4/wg1/en/faq-1-3.html)), because water is such an important greenhouse gas, the greenhouse effect is much larger in the humid tropics where the atmosphere has a high water content, than in the polar regions where the air is drier. Thus, when carbon dioxide is released into the atmosphere in small amounts in the polar regions where the air is drier, the effect on downward radiation is far greater than when a similar amount is released into the atmosphere in the tropics. Similarly, even a slight increase in water vapor in the upper atmosphere where the air is cold and dry, has a greater impact on the greenhouse effect that it would have when added to the lower atmosphere where the air is warm and moist.
Do Clouds Reduce or Enhance the Greenhouse Effect?
The answer to this question is not straight forward. Clouds form as a result of water vapor accumulating in the atmosphere, and as water vapor is a greenhouse gas, we can assume that they contribute to global warming. However, clouds also have reflective properties and contribute to the albedo affect mentioned earlier by reflecting some radiation away from the Earth. So, this currently remains a riddle that scientists are hard at work trying to resolve.

References and Image Credits
- Image Credit: By US EPA [Public domain], via Wikimedia Commons


Thanks somuch I have learnt a lot on how to stop global warming. I still have a questions,(1) how exactly does global warming affect rainfall partten?(2) what if we turn to the use of solar energy and stop burning fuels, can this reduce global warming?
i don’t have words to express how good work you have done what a wonderful website excellent work
1
Bring the change in you. Avoid plastics by all the means one could.
2
Plant as many trees and plants as your space permits.
3
Terrace gardens are a big advantage for people living in cities with limited garden space.
4
Contact your nearby clubs and conduct awareness classes in nearby schools and your locality.
5
Make a bigger move by joining hands with NGOs and local authorities to plant more trees.
6
Promote the usage of public transport to private vehicles.
7
Encourage people to use cycles
8
Conduct exhibitions in public halls that focuses on the increased natural calamities due to climate imbalance.
good awareness thoughts
Yours is the 1st website on my Google search for evidence to cite to people who don’t believe global warming is manmade. But I can’t use your site and will proceed to others instead. The organization that owns/runs the site is not listed nor are your funders. If you must be secret about these things, I guess you must. But I can’t use you without evidence that you are a legitimate, science-based group and are not funded by people who are partisan or who stand to somehow reap financial or political gain from activities that oppose fossil fuels. I look forward to your reply by email.
AFFORESTATION CAN ERADICATE BOTH GLOBAL WARMING CLIMATE CHANGE
Pingback: Global deal to limit use of HFCs a win in the battle against climate change - The Green Times